
Dimdima
Online Children's Magazine from India

Dimdima
Online Children's Magazine from India

Ebola Virus Disease (EVD) or simply Ebola, is an infectious and fatal disease. It is aused by four or five viruses of the Ebolavirus family. There is no treatment for the disease. Very few recover.
Ebola outbreaks occur primarily in remote villages in Central and West Africa. The virus is transmitted from wild animals to humans through contact with their blood and other bodily fluids. Wild animals that could be infected include monkeys, chimpanzees, antelopes, porcupines and fruit bats. The fruit bat is believed to be a natural host of the Ebola virus. It may harbour the virus without being affected by it. Once the virus has jumped to a human and he starts showing symptoms of the disease the virus can spread from him to others.
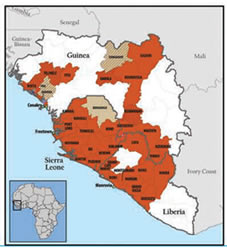 You cannot get Ebola from air, water or food. So far, Ebola cases have only shown up in Central and West Africa. The West African country of Liberia is one that is badly affected. Sierra Leone too is under threat from the virus.The rest of the world is keeping vigil at airports and other entry points to ensure that the disease doesn’t spread through travellers arriving from countries affected by the disease.
You cannot get Ebola from air, water or food. So far, Ebola cases have only shown up in Central and West Africa. The West African country of Liberia is one that is badly affected. Sierra Leone too is under threat from the virus.The rest of the world is keeping vigil at airports and other entry points to ensure that the disease doesn’t spread through travellers arriving from countries affected by the disease.
EXPLORE MORE...
Get Help or Give Help.
- Do you have a Science Question?
- Post it here and get the answer.
- Some questions posted by others are not yet answered.
- View those questions and answer them.
Dimdima is the Sanskrit word for ‘drumbeat’. In olden days, victory in battle was heralded by the beat of drums or any important news to be conveyed to the people used to be accompanied with drumbeats.
Bharatiya Vidya Bhavan
K. M Munshi Marg,
Chowpatty, Mumbai - 400 007
email : editor@dimdima.com
Bharatiya Vidya Bhavan
505, Sane Guruji Marg,
Tardeo, Mumbai - 400 034
email : promo@dimdima.com
Dimdima.com, the Children's Website of Bharatiya Vidya Bhavan launched in 2000 and came out with a Printed version of Dimdima Magazine in 2004. At present the Printed Version have more than 35,000 subscribers from India and Abroad.